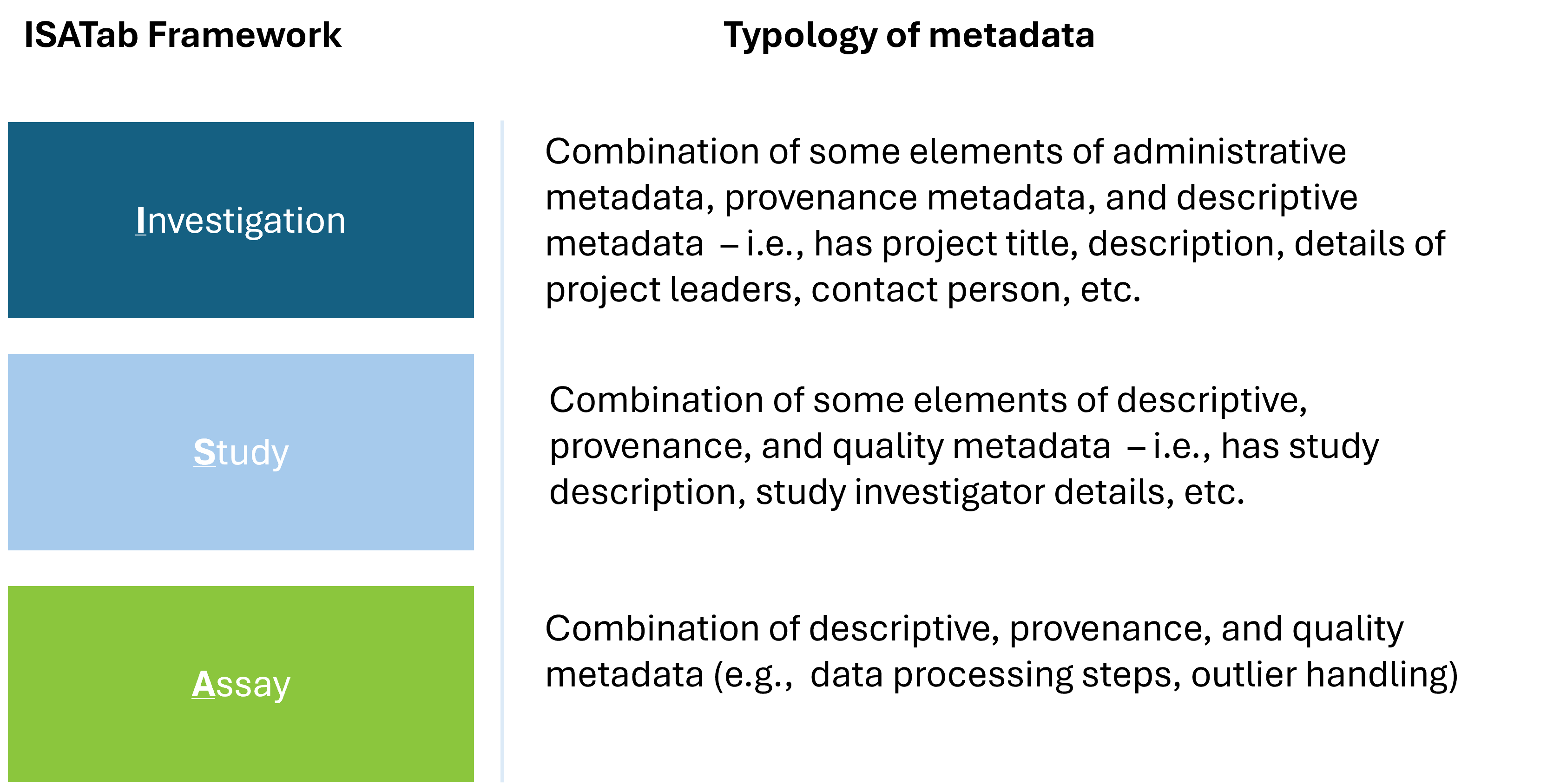The individual modules (i.e. sheets) of the excel workbook are given a name and the purpose of the sheets is explained below. There might be two or more analytical (measurement) endpoints to establish a primary endpoint, therefore ‘n’ refers to the number given to different assays (assays to measure LDH release, resazurin reduction, ATP levels can be given numbers as Assay-1, Assay-2, Assay-3 respectively for assessing the toxicological endpoint related to cell viability).
The Project module connects the studies within the specific WP5 project (i.e., via the project description) and other details, e.g., name of the project leader(s), Organisational affiliation of the Project Leader(s), name of the person who can be contacted.
The Action module is a unit of research and contains contextualising information about one or more assays.
The Assay module contains information about a set of assays, defined by the endpoint measured (e.g., increase in cytokine expression) and the technology employed (e.g. ELISA). The worksheet also describes the protocol followed and contains references to raw and processed data files (and their locations). There might be two or more analytical (measurement) endpoints to establish a primary endpoint, therefore additional assay files/excel sheets can be created for each of the different assays (such as, assays to measure LDH release, resazurin reduction, ATP levels for assessing endpoint related to cell viability).
The TestSubstance module contains overall information of the test chemical (s) tested in the project.
The PositiveControl module contains information on the positive controls used in the assay(s). (This sheet follows the descriptions provided in the TestSubstance module).
The NegativeControl module contains information on the negative controls used in the assay(s).
The AssaySummaryResult module contains the reporting template for the summary data to be reported for a particular assay. For additional assays, additional worksheets are suggested to used with sheet tab name as summary-assay-1, etc.
Field Type:
- Mandatory (M): The metadata element shall be documented. These fields are necessary for findability, provenance, proper data analysis, and reuse. Please ensure all information is entered accurately.
- Recommended (R): These properties are strongly suggested to be included in order to provide more insight into your meta(data).
- Conditionally required (C): The metadata element shall be documented if a previous element has been documented/recorded
- Optional (O): Provided to allow users to document their data in an enriched manner. Leave it blank only if it is very challenging to provide a response to a particular field. It is advised to provide as much detail as possible for completeness, QC/QA, and reproducibility.
This document is a genetic template in the sense that some of the fields may not be applicable to all toxicological experiments (e.g., pre-treatment, cell defining features), and in some cases, additional fields (e.g., differentiation status of cells in the case of immunotoxicity experiments) may be considered. Hence, the guide to fill the fields will be customised as per the (meta)data reporting template. Some information is routinely reported in publications, whereas some others are non-formalised (e.g., measured concentrations, lot/batch number) part of experiments, and hence generally not reported. Within each tab of the excel sheet, some of the fields (e.g., positive control, additives) are part of the general set-up of experiments and hence the way the information is asked for when filling it could be tedious.
Varied efforts exist (some in parallel) regarding toxicity meta(data) and consistency in definitions is yet to be achieved, therefore, we have specified the meaning of several terms while describing the field names, and wherever they have been taken from published resources (or where ontologies exist, even if overlapping efforts are there, e.g., Foundation Model of Anatomy and UBERON for anatomy) the reference / and ontology source of the chosen term is provided within the description provided in the guidance document.
Each of the sheets in the (meta)data reporting template encompasses descriptive, administrative, quality, and provenance metadata types. See Figure 3.1 below which is a snapshot of these different metadata types.
Investigation (i.e.) Project sheet
| TitleOfProject |
| Description |
Title of the project |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Hazard characterisation of the mycotoxins enniatins and Alternaria toxins in order to close data gaps and improve risk assessment for human health |
| PARCProjectID |
| Description |
The Project ID as used by WP5 |
| Field Type |
Mandatory |
| Data Type |
String |
| Examples |
P5.1.1.a_Y1_Toxins_BfR_UNIVIE (select from dropdown) |
| ProjectAcronym |
| Description |
Acronym of the project |
| Field Type |
Optional |
| Data Type |
Text |
| Examples |
Toxins |
| DescriptionOfProject |
| Description |
Short summary of the project (up to 500 words) which describes its aims/objectives and the reasons for doing the project. |
| Field Type |
Mandatory |
| Data Type |
Text |
| DescriptionOfProjectPARCwebsite |
| Description |
Project descriptions on PARC website (will be later in the form of nanopublications, and the link will be provided here). |
| Field Type |
Recommended |
| Data Type |
URL |
| FullNameProjectLeader1 |
| Description |
Project Leader’s full name. The names person(s) who conceptualised the project, responsible for drafting the project description, and responsible for its delivery.
In the case of some PARC projects, project leader and project manager / deputy manager could be the same person |
| Field Type |
Mandatory |
| Data Type |
Text |
| EmailProjectLeader1 |
| Description |
Email id of the project leader |
| Field Type |
Mandatory |
| Data Type |
Text |
| InstitutionalAffiliationProjectLeader1 |
| Description |
The host institution of the project leader. |
| Field Type |
Mandatory |
| Data Type |
Text |
| RORIdentifier1 |
| Description |
The persistent identifier (PID) of the institution as provided in the Research Organisation Registry. Refer to https://ror.org/. If the PID is unavailable for the Institution, mention “Not Available” |
| Field Type |
Mandatory |
| Data Type |
URL |
| Examples |
https://ror.org/02hssy432 |
| CountryOfInstitution1 |
| Description |
Name of the country where the Institution’s office is located to which the project leader is associated with, |
| Field Type |
Mandatory |
| Data Type |
Text |
| ContactPointP |
| Description |
Full name of the person who can be contacted post project end regarding the research results (e.g. access, troubleshoot, or respond to questions/ issues related to the research). It could be the same as project leader (s)
If there is no single person who can be contacted, but a central/generic ID is provided, mention “Not available” |
| Field Type |
Mandatory |
| Data Type |
Text |
| ContactPointPEmail |
| Description |
Email id of the Contact Point |
| Field Type |
Mandatory |
| Data Type |
String |
| HostingInstitution |
| Description |
Typically, the organisation allowing the resource to be available on the
internet through the provision of its hardware/software/operating support.
Note: May also be used for an organisation that stores the data offline. Often a data centre (if that data centre is not the “publisher” of the resource.)
Ref: DateCite https://schema.datacite.org/meta/kernel-4.1/doc/DataCite-MetadataKernel_v4.1.pdf
This question is also in PARC DSW Data Management Plan tool. |
| Field Type |
Optional |
| Data Type |
Text |
| Examples |
University of Aveiro’s - Research Data Repository
https://www.ua.pt/pt/sbidm/dunas |
| Repository |
| Description |
The name of the repository where the dataset from the project will be deposited. If not known, discuss with Indrani (i.mahapatra@bham.ac.uk)
This question is also in PARC DSW Data Management Plan tool. |
| Field Type |
Recommended |
| Data Type |
Text |
| RepositoryWeblink |
| Description |
The weblink of the chosen repository where the dataset will be deposited (if not yet known discuss with Indrani)
This question is also in PARC DSW Data Management Plan tool. |
| Field Type |
Conditionally required |
| Data Type |
URL |
| Publication |
| Description |
The title of the publication(s) from the study. If the study associated with the dataset has not been published, leave blank.
This question is also in PARC DSW Data Management Plan tool. |
| Field Type |
Recommended |
| Data Type |
Text |
| PublicationLink |
| Description |
The weblink to the publication.
This question is also in PARC DSW Data Management Plan tool. |
| Field Type |
Conditionally required |
| Data Type |
URL |
| Deliverable |
| Description |
The full deliverable (s) name (with number) which will contain the information about the project/study.
This question is also in PARC DSW Data Management Plan tool. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
D5.4 :Inventory of omics data, biochemical data, other relevant readouts in public domain repositories for all PARC selected adverse outcomes |
| DescriptionOfDataGeneration |
| Description |
Explain the datasets that would be generated from the studies
Dataset: Organised collection of data or objects in a computational format, that are generated or collected by researchers in the course of their investigations, regardless of their form or method, that form the object on which researchers test a hypothesis. This includes the full range of data: raw, unprocessed datasets, proprietary generated and processed data and secondary data obtained from third parties. Ref: https://terms.codata.org/rdmt/dataset
This question is also in PARC DSW Data Management Plan tool. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Data related to relative (fold change) in hormone levels at each concentration, normalised metabolite ratio peak data per sample |
Action (activity/study) sheet
The last updated date and release date are metadata fields more applicable to the repository where the data is deposited and is not fully the responsibility of the WP5 researcher generating the data. They have been included here for the sake of completeness.
| StudyDetails |
| Description |
Brief description of the study. For in vitro studies, 1) describe relevance of test system to tissue/organ/species of interest, 2) Provide details of the mechanistic understanding, and 3) elaborate on the defining features of the test system, if applicable.
Provide necessary background information for the end user to understand the rationale for why the study was undertaken, including the regulatory question(s), if any.
According to ISA-tab, study is a unit of research; and “contains contextualising information for one or more Assay”.
Ref: https://isa-specs.readthedocs.io/en/latest/isamodel.html#study |
| Field Type |
Mandatory |
| Data Type |
Text |
| MethodNameID |
| Description |
The method name will be included based on the naming convention of the methods / assays decided by WP5. The method name can be modelled in a manner that it includes the project acronym, organisation generating the data, test system, toxicity domain, biological endpoint, exposure, unique ID of the experiment, etc. |
| Field Type |
Recommended |
| Data Type |
String |
| Examples |
BPA_HumTox_BfR_HepG2_Carcinogenicity_genotoxicity_comet_24h_ExperimentID |
| ToxicityDomain |
| Description |
The main organ or organ system that is being explored for toxic impacts |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
carcinogenesis, hepatotoxicity, renal toxicity, neurotoxicity, reproductive toxicity, endocrine toxicity, immunotoxicity, developmental toxicity |
| ToxicologicalEndpoint |
| Description |
Toxicological endpoint is more specific when compared to toxicity domain. .
A direct marker of progression to an adverse outcome - e.g., morphological or physiological changes, functional impairments, disease symptoms or death - used to describe an adverse health effect (or a probability of that adverse effect) resulting from exposure to a test item. The test system response to an exposure of a test item may be measured by a series of endpoints. The most sensitive endpoint (critical endpoint) is the one that occurs at the lowest exposure level and associated with an adverse response (committed step)
(ref: GIVIMP Second edition) |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Cytotoxicity/cell death, cell proliferation, oxidative stress, membrane depolarisation, cytokine expression, differential gene expression etc. |
| TypeOfTestSystem |
| Description |
A test system is any biological, chemical or physical system or a combination thereof used in a study: ref: OECD (2018), Guidance Document on Good In Vitro Method Practices (GIVIMP))
Examples of physical chemical-based test systems: serum protein, peptide, enzyme.
Select complex biological test system for example in case of: 3D model, induced pluripotent stem cells, organ on a chip, co-cultures, etc.
Select from dropdown ‘other:’ in case you don’t find a suitable option, for example when your test system is a test kit or a lower in vivo organism.
Selection from the dropdown in Excel. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Organism |
| Description |
A living entity.
(Alternate definition: A living thing, such as an animal, a plant, a bacterium, or a fungus.; Any individual living (or previously living) being).
Ref: https://ontobee.org/ontology/NCIT?iri=http://purl.obolibrary.org/obo/NCIT_C14250
Common name for the provided NCBI taxon ID. Latin name of species can be used, if appropriate. |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
Human, Caenorhabditis elegans, Danio rerio, Daphnia magna, Mus musculus |
| CellType |
| Description |
Cell Type is advised to be described as per resources, such as atlases/Pan_Immune_CellTypist/v2/encyclopedia/encyclopedia_table.xlsx of the CellTypist (for Immune cells)
HuBMAP Common Coordinate Framework (CCF) ASCT+B tables (e.g. https://apps.humanatlas.io/kg-explorer/ for cell types as per anatomical structures)
Cell Ontology (CL) https://www.ebi.ac.uk/ols/ontologies/cl.
Be as specific as possible. |
| Field Type |
Recommended |
| Data Type |
Text, URL |
| Examples |
Granulocytes, epithelial, endothelial, astrocyte |
| CellLine |
| Description |
A cell line is defined as a genetically stable and homogenous population of cultured cells that shares a common propagation history (i.e., has been successively passaged together in culture).
Ref: Sarntivijai, Sirarat, et al. 2014. “CLO: The Cell Line Ontology.” Journal of Biomedical Semantics 5(1):37. doi: 10.1186/2041-1480-5-37. |
| Field Type |
Mandatory |
| Data Type |
Text |
| CellLineGroup |
| Description |
The replication capability of the cell and/or cell line |
| Field Type |
Optional |
| Data Type |
Text |
| Examples |
finite, continuous, immortalised, stem cell lines |
| CellLineDesignation |
| Description |
The identification provided when the test systems is sourced from recognised cell culture banks (e.g. ATCC, ECACC, DMSZ, UKSCB). Cell lines can have different designation when cultured in house when compared to cells sourced from external cell culture banks. |
| Field Type |
Recommended |
| Data Type |
Text |
| Heterogenicity |
| Description |
Culturing conditions of cells |
| Field Type |
Recommended |
| Data Type |
Text |
| CellCultureModel |
| Description |
Cell culture model in vitro |
| Field Type |
Recommended |
| Data Type |
Text |
| AssayName |
| Description |
An assay is a defined laboratory procedure for qualitatively or quantitatively measuring the presence or amount or the functional activity of a target or analyte.
Ref: OECD (2018), Guidance Document on Good In Vitro Method Practices (GIVIMP).
Mention the assay name here (with branding details, if commercially procured). If the assay is described in a publication, provide the DOI. |
| Field Type |
Mandatory |
| Data Type |
Text; doi |
| AssayThroughput |
| Description |
The number of samples, conditions, or compounds that an assay can process within a defined period. |
| Field Type |
Recommended |
| Data Type |
Text |
| TypeOfExposure |
| Description |
The number of applications of the exposure dose |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Single dose, multiple doses |
| TypeOfToxicityStudy |
| Description |
Based on duration of exposure, toxicity can be categorised into four levels : Acute, Chronic, Sub-chronic, sub-acute |
| Field Type |
Recommended |
| Data Type |
Text |
| ExposureDuration |
| Description |
The total time of exposure to the test substance |
| Field Type |
Mandatory |
| Data Type |
Integer |
| Examples |
24, 48, 7 |
| UnitofExposureDuration |
| Description |
Unit of exposure time/duration |
| Field Type |
Mandatory |
| Data Type |
text |
| Examples |
hrs, d |
| ExposureConcentrationsApplied |
| Description |
The test substance concentration within the exposure matrix (e.g., water, media) used in the exposure scheme. Can be provided as a range with minimum and maximum concentrations. |
| Field Type |
Mandatory |
| Data Type |
Numeric |
| ExposureConcentrationsAppliedUnit |
| Description |
Unit of the test substance’s actual concentration for conducting the exposure experiments, preferably molar concentrations (e.g. microM) or ppm. Please do not use special characters like µ to avoid mojibake. |
| Field Type |
Mandatory |
| Data Type |
Text |
| ExposureConcentrationActual |
| Description |
The concentration of the substance in the exposure medium, measured analytically at specific time points during the study. If measured concentrations are not available, please mention “not measured”. |
| Field Type |
Recommended |
| Data Type |
Numeric/text |
| ExposureConcentrationActualUnit |
| Description |
Unit of the test substance’s measured/actual concentration, preferably molar concentrations (e.g. microM) or ppm. Please do not use special characters like µ to avoid mojibake. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Endpoint1definition |
| Description |
Explain the biochemical process, response or effect assessed in the test system.
Describe how the measured signal corresponds to a direct read out of the analyte (e.g. enzyme activity) or an indirect complex response such as cell survival, proliferation, localization of a protein, nuclear translocation. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Endpoint1 measure |
| Description |
The biological or chemical process or response assessed in the test system |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Measurement of extracellular LDH, reduction of resazurin by healthy cells, measurement of ATP levels, using PI to stain DNA of cells |
| Endpoint1readoutmethod |
| Description |
Explain the technology that is being used to detect change? |
| Field Type |
Mandatory |
| Data Type |
text |
| Endpoint1normalisation |
| Description |
Describe how the endpoint normalised, if applicable. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Percent of vehicle control |
| TestSubstance |
| Description |
Common/widely used name of the chemical/substance being tested, i.e., the test item. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Bisphenol Z |
| StudyLead |
| Description |
The person responsible for supervising/managing a sub-unit of a PARC project. In case of PARC, they are generally, the activity/use case lead, and could be deputy managers of the projects. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Joanna Smith |
| ORCIDStudyLead |
| Description |
Provide the ORCID ID of the study lead |
| Field Type |
Optional |
| Data Type |
String |
| EmailStudyLead |
| Description |
Provide the email id of the study lead |
| Field Type |
Mandatory |
| Data Type |
Text |
| RoleStudyLead |
| Description |
Describe the role of the Study Lead(s) (within the project) to establish provenance. Only choose one that best describes the role of the Study Lead (even if not comprehensively) below:
Supervision role: Oversight and leadership responsibility for the research activity planning and execution, including mentorship external to the core team.
Project administration role: Management and coordination responsibility for the research activity planning and execution.
Coordination role: The organization of the different elements needed to implement a study.
Conceptualization role: Ideas; formulation or evolution of overarching research goals and aims |
| Field Type |
Recommended |
| Data Type |
Text |
| InstitutionalAffiliationStudyLead |
| Description |
The name of the institution to which the study lead is associated with. |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
University of Leiden |
| RORIdentifierStudyLead |
| Description |
The identifier of the research institution in the Research Organization Registry (ROR) that the study lead is affiliated with. |
| Field Type |
Recommended |
| Data Type |
URL |
| Examples |
027bh9e22 |
| SummaryDatafileReference |
| Description |
Name of the file with effects results from the experiments. |
| Field Type |
Recommended |
| Data Type |
String |
| Examples |
Stringsummary.csv; stringsummary.xlsx |
| Researcher |
| Description |
The role a person has of being a researcher involved in a research investigation, for example by conducting experiments, collecting and analysing data, or undertaking formal study (ref: http://purl.org/spar/scoro/researcher)
In addition to credit and provenance, it helps in quality control and data tracking. Provide full name of the person, preferably as registered in ORCID registry, or name is scientific publications if any, or professional name most widely used. |
| Field Type |
Mandatory |
| Data Type |
text |
| Examples |
Jane Doe |
| ResearcherORCIDiD |
| Description |
The ORCID iD of the researcher who is generating the data in the laboratory. |
| Field Type |
Mandatory |
| Data Type |
String/integer |
| ContactSOFullName |
| Description |
The name of the person who can be contacted for acquiring knowledge about the study, or acquisition of data/resource, if available. Could be same as Study lead email id or researcher email. |
| Field Type |
Mandatory |
| Data Type |
text |
| Examples |
John Smith |
| ContactSOEmail |
| Description |
The email id of the point of contact who can be contacted for acquiring knowledge about the study or acquisition of the data/resource. Could be same as Study lead email id or researcher email. |
| Field Type |
Mandatory |
| Data Type |
Text |
| DateCreation |
| Description |
Date on which the template was completed to be sent to CRAhub for registration of the metadata. |
| Field Type |
Recommended |
| Data Type |
Date: YYY-MM-DD (ISO 8601, date and time format, i.e., YYYY-MM-DD) |
| DepositedDate |
| Description |
Date on which the data was sent to be uploaded in the selected public repository. |
| Field Type |
Recommended |
| Data Type |
Date: YYY-MM-DD (ISO 8601, date and time format, i.e., YYYY-MM-DD) |
| ReleaseDate |
| Description |
The date on which the data is expected to be publicly available (include embargo period, if any) |
| Field Type |
Recommended |
| Data Type |
YYYY-MM-DD |
| LastUpdatedDate |
| Description |
The date when the dataset was examined/re-examined and revised or amended. |
| Field Type |
Optional |
| Data Type |
YYYY-MM-DD |
| DescriptionOfDataGeneration |
| Description |
Explain the datasets that would be generated from the studies
Dataset: Organised collection of data or objects in a computational format, that are generated or collected by researchers in the course of their investigations, regardless of their form or method, that form the object on which researchers test a hypothesis. This includes the full range of data: raw, unprocessed datasets, proprietary generated and processed data and secondary data obtained from third parties. Ref: https://terms.codata.org/rdmt/dataset
This question is also in PARC DSW Data Management Plan tool. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Data related to relative (fold change) in hormones level at each concentration, normalized metabolite ratio peak data per sample |
Test Substance Sheet
| TestSubstance |
| Description |
Common/widely used name of the chemical/substance being tested, i.e., the test item. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Bisphenol Z |
| TestSubstanceID |
| Description |
An identifier given in the project. This will be via PID generators or nanopublications |
| Field Type |
Mandatory |
| Data Type |
String |
| ChemicalName |
| Description |
Full chemical name as per IUPAC. If the test material/test substance doesn’t have any stoichiometric name or is a complex mixture or unknown or variable composition mention “NotAvailable” |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
4,4′-(1,1-Cyclohexanediyl)diphenol |
| TestSubstanceDescription |
| Description |
If the ChemicalName field is “NotAvailable”, describe about the test substance and method of preparation briefly. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| CASRegistryName |
| Description |
The name of the chemical/substance in the CAS Registry. Mention as “NotAvailable” if no registry name; or if it is the same as chemical name, leave this field blank |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
1,1-Bis(4-hydroxyphenyl)cyclohexane |
| CASRegistryNumber |
| Description |
The unique numeric identifier for the test substance if available in the CAS registry |
| Field Type |
Conditionally required |
| Data Type |
String |
| Examples |
843-55-0 |
| TestSubstanceECRefNo |
| Description |
The unique identifier assigned to substances for regulatory purposes within the European Union by the European Commission. |
| Field Type |
Optional |
| Data Type |
String |
| Examples |
212-677-1 |
| PubChemCID |
| Description |
Provide the identifier from the PubChem Database |
| Field Type |
Recommended |
| Data Type |
Integer |
| Examples |
2244 |
| SMILES |
| Description |
The SMILE (Simplified Molecular-Input Line-Entry System) string for the test substance. Use PubChem website as first instance for the SMILE, if not available in PubChem, then check CAS Common Registry, and then ECHA’s chemical database. |
| Field Type |
Recommended |
| Data Type |
String |
| Examples |
OC1=CC=C(C2(CCCCC2)C3=CC=C(O)C=C3)C=C1 |
| InChI |
| Description |
The “International Chemical Identifier” for the test substance |
| Field Type |
Recommended |
| Data Type |
String |
| Examples |
InChI=1S/C18H20O2/c19-16-8-4-14(5-9-16)18(12-2-1-3-13-18)15-6-10-17(20)11-7-15/h4-11,19-20H,1-3,12-13H2 |
| InChIKey |
| Description |
The hashed version of InChI |
| Field Type |
Optional |
| Data Type |
String |
| Examples |
SDDLEVPIDBLVHC-UHFFFAOYSA-N |
| ChemicalSupplier |
| Description |
The name of the company from whom the test substance was purchased/procured. If synthesised in the laboratory, mention “in-house”. If by a partner organisation, provide the name of the organisation (e.g. Joint Research Centre). |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Joint Research Centre (EC), Sigma-Aldrich |
| ProductNumber |
| Description |
The product number/catalogue number of the test substance as used in the study |
| Field Type |
Optional |
| Data Type |
String |
| Examples |
450421 |
| PortfolioBrand |
| Description |
Provide the name of the portfolio brand, if any |
| Field Type |
Optional |
| Data Type |
Text |
| Examples |
Supelco of Sigma-Aldrich |
| BatchNumber |
| Description |
The batch number of the test substance. If not available, mention “NotAvailable” |
| Field Type |
Mandatory |
| Data Type |
String |
| ChemicalForm |
| Description |
The physical form of the test compound (powder, crystals, in solution) |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Powder |
| ChemicalFormDescription |
| Description |
The solution in which the compound was purchased and its concentration, if the compound was procured in solution form |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
in methanol, 1 mg/ml |
| ChemicalPurity |
| Description |
The percentage purity of the test substance. Provide quantitative information. Qualitative information such as “reagent grade” is not considered sufficient. Do not add percentage symbol. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
≥99.9 |
| StorageTemperature |
| Description |
The temperature at which the test substance is stored, in °C |
| Field Type |
Mandatory |
| Data Type |
Number |
| SolventName |
| Description |
Chemical name of the solvent to be used to dissolve the test substance |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
Dimethyl sulfoxide (DMSO) |
| SolventSupplier |
| Description |
Name of the company who manufactures or supplied the solvent |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
Sigma-Aldrich |
| SolventProductNumber |
| Description |
The catalogue number/product number of the solvent |
| Field Type |
Recommended |
| Data Type |
String |
| Examples |
D8418 |
| SolventBatchNumber |
| Description |
Lot/batch number of the solvent |
| Field Type |
Recommended |
| Data Type |
String |
| SolventPurity |
| Description |
Report the purity in percentage based on information available during procurement of the solvent |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
≥99.9 |
| MethodOfDissolution |
| Description |
The method adopted to dissolve the test substance in the solvent |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
vortexing, sonification |
| StockSolutionConcentration |
| Description |
The concentration of the stock solution (molar concentrations - mM, microM, etc. - preferred). Please do not use special characters like µ to avoid mojibake. |
| Field Type |
Mandatory |
| Data Type |
Number |
| StockSolutionUnit |
| Description |
The concentration unit of the stock solution, if other than molar concentrations |
| Field Type |
Mandatory |
| Data Type |
Text |
| StockAliquotStorageConditions |
| Description |
Detail the storage conditions of the stock aliquot, if stock is not prepared fresh before an experiment |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
Made fresh on day or made and stored at temperature 4 degree Celsius for maximum 1 month |
Positive Control Sheet
| PCcommon name |
| Description |
Common/widely used name of the chemical/substance used as positive control |
| Field Type |
Mandatory |
| Data Type |
Text |
| PCChemicalName |
| Description |
Full chemical name as per IUPAC, if available. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| PCDescription |
| Description |
Briefly describe why this substance is chosen as positive control. |
| Field Type |
Mandatory |
| Data Type |
Text |
| PCCASRegistryName |
| Description |
The name of the substance used as positive control, if available, in the CAS Registry. |
| Field Type |
Conditionally Required |
| Data Type |
Text |
| PCCASRegistryNumber |
| Description |
The unique numeric identifier for the substance used as positive control if available in the CAS registry |
| Field Type |
Conditionally required |
| Data Type |
String |
| PCSMILES |
| Description |
The SMILE (Simplified Molecular-Input Line-Entry System) string for the substance used as positive control |
| Field Type |
Optional |
| Data Type |
String |
| PCInChI |
| Description |
The “International Chemical Identifier” for the substance used as positive control |
| Field Type |
Optional |
| Data Type |
String |
| PCInChIKey |
| Description |
The hashed version of InChI |
| Field Type |
Optional |
| Data Type |
String |
| Examples |
SDDLEVPIDBLVHC-UHFFFAOYSA-N |
| PCSupplier |
| Description |
The name of the company from whom the substance used as positive control was purchased/procured. |
| Field Type |
Recommended |
| Data Type |
Text |
| PCProductNumber |
| Description |
The product number/catalogue number of the test substance as used in the study |
| Field Type |
Optional |
| Data Type |
String |
| PCPortfolioBrand |
| Description |
Provide the name of the portfolio brand, if any |
| Field Type |
Optional |
| Data Type |
Text |
| PCBatchNumber |
| Description |
The batch number of the substance used as positive control. If not available, mention “Not Available” |
| Field Type |
Recommended |
| Data Type |
String |
| PCForm |
| Description |
The physical form of the substance used as positive control (powder, crystals, in solution) |
| Field Type |
Recommended |
| Data Type |
Text |
| PCFormDescription |
| Description |
The solution in which the compound was purchased and its concentration, if the compound was procured in solution form. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| PCPurity |
| Description |
The percentage purity of the substance used as positive control. Provide quantitative information. Qualitative information such as “reagent grade” is not considered sufficient. Do not add percentage symbol. |
| Field Type |
Recommended |
| Data Type |
Text |
Negative Control Sheet
| NCcommon name |
| Description |
Common/widely used name of the chemical/substance used as positive control |
| Field Type |
Mandatory |
| Data Type |
Text |
| NCChemicalName |
| Description |
Full chemical name as per IUPAC, if available. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| NCDescription |
| Description |
Briefly describe why this substance is chosen as negative control. |
| Field Type |
Recommended |
| Data Type |
Text |
| NCCASRegistryName |
| Description |
The name of the substance used as negative control, if available, in the CAS Registry. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| NCCASRegistryNumber |
| Description |
The unique numeric identifier for the substance used as negative control if available in the CAS registry |
| Field Type |
Conditionally required |
| Data Type |
String |
| NCSMILES |
| Description |
The SMILE (Simplified Molecular-Input Line-Entry System) string for the substance used as negative control |
| Field Type |
Optional |
| Data Type |
String |
| NCInChI |
| Description |
The “International Chemical Identifier” for the substance used as negative control |
| Field Type |
Optional |
| Data Type |
String |
| NCInChIKey |
| Description |
The hashed version of InChI |
| Field Type |
Optional |
| Data Type |
String |
| Examples |
SDDLEVPIDBLVHC-UHFFFAOYSA-N |
| NCSupplier |
| Description |
The name of the company from whom the substance used as negative control was purchased/procured. |
| Field Type |
Recommended |
| Data Type |
Text |
| NCProductNumber |
| Description |
The product number/catalogue number of the test substance as used in the study |
| Field Type |
Optional |
| Data Type |
String |
| NCPortfolioBrand |
| Description |
Provide the name of the portfolio brand, if any |
| Field Type |
Optional |
| Data Type |
Text |
| NCBatchNumber |
| Description |
The batch number of the substance used as negative control. If not available, mention “Not Available” |
| Field Type |
Recommended |
| Data Type |
String |
| NCForm |
| Description |
The physical form of the substance used as negative control (powder, crystals, in solution) |
| Field Type |
Recommended |
| Data Type |
Text |
| NCFormDescription |
| Description |
The solution in which the compound was purchased and its concentration, if the compound was procured in solution form |
| Field Type |
Conditionally required |
| Data Type |
Text |
| NCPurity |
| Description |
The percentage purity of the substance used as negative control. Provide quantitative information. Qualitative information such as “reagent grade” is not considered sufficient. Do not add percentage symbol. |
| Field Type |
Recommended |
| Data Type |
Text |
Assay Sheet
| Experiment_ID |
| Description |
The experiment ID – the syntax to be decided by WP5. Can be extrapolated from the the MethodNameID. Used for internal reference. |
| Field Type |
Recommended |
| Data Type |
String |
| CellType |
| Description |
Describe as per the following resources: atlases/Pan_Immune_CellTypist/v2/encyclopedia/encyclopedia_table.xlsx of the CellTypist
HuBMAP Common Coordinate Framework (CCF) ASCT+B tables
Cell Ontology. |
| Field Type |
Recommended |
| Data Type |
Text, url |
| Examples |
Granulocytes, epithelial |
| CellLine |
| Description |
A cell line is defined as a genetically stable and homogenous population of cultured cells that shares a common propagation history (i.e. has been successively passaged together in culture). Ref: Sarntivijai, Sirarat, et al. 2014. “CLO: The Cell Line Ontology.” Journal of Biomedical Semantics 5(1):37. doi: 10.1186/2041-1480-5-37. |
| Field Type |
Mandatory |
| Data Type |
Text, URL |
| Examples |
HepG2, A549, hiPS - Human induced-pluripotent stem cells, 293FT,Human embryonic stem cell (hESC) |
| CellLineOntologyID |
| Description |
Cell Line Ontology
Ref: https://www.ebi.ac.uk/ols4/ontologies/clo |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
CLO:0050126 for RCB0027 cell which is an immortal human cell line cell that has the characteristics: Chronic myelogenous leukemia, sensitive to NK cell. Differentiate to erythroid cells |
| CellLineGroup |
| Description |
The replication capability of the cell line |
| Field Type |
Optional |
| Data Type |
Text |
| Examples |
finite, continuous, immortalized, stem cell lines |
| CellProvider |
| Description |
Name of the company or lab or repository providing the cells |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Invitrogen, ThermoFisher, ECACC |
| CellCatalogueNumber |
| Description |
The unique identifier of a cell line (e.g., ATCC, ECACC, DMSZ, UKSCB designations). |
| Field Type |
Recommended |
| Data Type |
String |
| Examples |
84101003, HB-8065, R70007, PCS-450-012 |
| CellBatchNumber |
| Description |
Provide the batch number of the cells procured. |
| Field Type |
Mandatory |
| Data Type |
String |
| CellLineDesignation |
| Description |
The identification provided when the test systems is sourced from recognised cell culture banks (e.g. ATCC, ECACC, DMSZ, UKSCB. Cell lines can have different designation when cultured in house and not procured from external cell culture bank. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
HeLa, CCL-2,Hep G2 [HEPG2] |
| CellLlineDesignationRRID |
| Description |
Research Resource ID (RRID) from Cellosaurus (https://www.cellosaurus.org/index.html) |
| Field Type |
Optional |
| Data Type |
String |
| Examples |
RRID:CVCL_0030,RRID:CVCL_0027 |
| CultureMediumComposition |
| Description |
Describe the cell culture medium composition briefly with key components, such as serum type and percentage, antibiotics. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Roswell Park Memorial Institute Medium (RPMI 1640) supplemented with 10 % (v/v) Fetal Bovine Serum (FBS) heat-inactivated, 100 U/mL penicillin, 100 μg/mL streptomycin, and 4 mM/L glutamine |
| CellMediumSupplierCatalogueNumber |
| Description |
Name of the company from which medium was procured along with catalogue number |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
ThermoFisher (11965 - DMEM, high glucose) |
| SerumPercentage |
| Description |
Percentage of serum used in the cell culture media, if used. Avoid adding percentage symbol after reporting the data. |
| Field Type |
Conditionally required |
| Data Type |
Numeric |
| SerumSource |
| Description |
Serum supplier |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
ThermoFisher Scientific |
| SerumLotNumber |
| Description |
Provide the lot/control number for the serum |
| Field Type |
Recommended |
| Data Type |
String |
| HeatInactivation |
| Description |
Mention in only “no or yes” as a response whether serum was subjected to heat |
| Field Type |
Mandatory |
| Data Type |
Text |
| MediumAdditive1 |
| Description |
Mention the additives to the cell culture medium. |
| Field Type |
Recommended |
| Data Type |
String |
| MediumAdditive1_supplier |
| Description |
Provide the name of the supplier providing the additive and catalogue number associated with it. |
| Field Type |
Recommended |
| Data Type |
String |
| MediumAdditive1_ concentration |
| Description |
Provide the concentration of the additive used in the experiment, preferably in molar concentrations. |
| Field Type |
Recommended |
| Data Type |
Numeric |
| MediumAdditive1_concentrationUnit |
| Description |
The measurement unit used to express the concentration or amount of the additive |
| Field Type |
Recommended |
| Data Type |
Text |
| OtherMaterialsAdded1 |
| Description |
Other materials (e.g. antibiotics) added to the cell culture media |
| Field Type |
Optional |
| Data Type |
Text |
| OtherMaterialsAdded1Concentration |
| Description |
Provide the concentration of the material added in the experiment, preferably in molar concentrations |
| Field Type |
Optional |
| Data Type |
Numeric |
| OtherMaterialsAdded1Unit |
| Description |
The measurement unit used to express the concentration or amount of the added material |
| Field Type |
Optional |
| Data Type |
Text |
| MicrowellPlateFormat |
| Description |
Number of wells in the microwell plate used for the exposure experiment |
| Field Type |
Mandatory |
| Data Type |
Integer |
| Examples |
48, 96, 384 or 1536 |
| MicrowellPlateDesign |
| Description |
Details of the microwell plate design (i.e. describe colour and shape of microwell plates). |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
transparent, black, white, flat, round bottom |
| PlateManufacturer |
| Description |
Name of the plate manufacturer |
| Field Type |
Recommended |
| Data Type |
Text |
| PlateProductNumber |
| Description |
The product number provided by the supplier for the microwell plate used in the experiment |
| Field Type |
Recommended |
| Data Type |
String |
| VolumeOfIncubationMediumWell |
| Description |
Amount of liquid growth media in each well |
| Field Type |
Mandatory |
| Data Type |
Numeric |
| VolumeOfIncubationMediumWellUnit |
| Description |
The measurement unit via which volume of media in each microwell is expressed. |
| Field Type |
Mandatory |
| Data Type |
Text |
| CellPassageNumber |
| Description |
The term “passage” refers to the transfer of cells from one culture vessel to another or a number of vessels, usually with the aim of increasing cell numbers (ref: Pamies et.al., 2022)
Passage number refers to the number of times the cell line has been re-plated (adherent cultures) or re-seeded (suspension cultures) since it was first established or thawed from a frozen stock. |
| Field Type |
Recommended |
| Data Type |
Integer |
| SeedingDensity |
| Description |
Number of cells placed on a growth material per unit area. |
| Field Type |
Recommended |
| Data Type |
Integer |
| SeedingDensityUnit |
| Description |
The measurement unit via which seeding density is expressed |
| Field Type |
Recommended |
| Data Type |
Text |
| CellCultureTechnique |
| Description |
Describe how the cells were cultured in the medium. (or just give cell growth mode – adherent/suspension) |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
2D- Adherent (petri dish or flask) 3D-Suspension cultures on non-adherent plates, 3D-Cultures in concentrated medium or in gel-like substances, 3D-Cultures on scaffold, 2D-suspension, air-liquid interface, in transwell inserts etc. |
| CellPretreatment |
| Description |
Were the cells exposed to any specific substances before exposure with the test substance? Respond in “yes” and “no” |
| Field Type |
Mandatory |
| Data Type |
Text |
| PretreatmentReasons |
| Description |
Describe why was pre-treatment done. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
To remove antibiotics |
| PretreatmentCompound |
| Description |
Full name of the chemical compound(s) used in pre-treatment |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
phorbol 12-myristate-13-acetate |
| DilutionType |
| Description |
The dilution technique followed in the experiment |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
Direct, Serial, Logarithmic |
| DilutionSteps |
| Description |
Provide information on the dilution steps from ‘stock solution’ including the final % of vehicle/solvent in the exposure medium |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
The test material was first diluted in 70% ethanol and subsequently diluted 500-fold in cell culture medium. Another 2-fold dilution was executed in the well to obtain a total of 1000-fold dilution and a final solvent concentration of 0.07%. (ref: OHT201) |
| AnalyticalEndpoint1definition |
| Description |
Explain the method which helps to quantify the toxicological endpoint. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
If cell viability is the toxicological endpoint, then one of the analytical methods to assess cell viability is measuring LDH-release. |
| AssayName |
| Description |
A defined laboratory procedure for qualitatively or quantitatively measuring the presence or amount or the functional activity of a target or analyte. An assay can be considered as a technical operation that consists of determination of one or more characteristics of a given product, process or service according to a specified procedure.
Ref: OECD (2018), Guidance Document on Good In Vitro Method Practices (GIVIMP).
Mention the assay name here (with branding details, if commercially procured). If the assay is described in a publication, provide the DOI. |
| Field Type |
Mandatory |
| Data Type |
Text, doi: |
| Examples |
IL-2 Luc Assay,LDH Glo Cytotoxicity Assay |
| AssayKit |
| Description |
Name of the commercially procured assay kit used to perform the assay. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
ATP Assay Kit, Pierce BCA Protein Assay Kit |
| AssayKitProvider |
| Description |
Name of the company which manufactures (markets/distributes) the assay kit, with as much details as possible |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
Cambridge Bioscience (manufacturer: Cell Biolabs, Inc.), Thermo Scientific (part of Thermo Fisher Scientific) |
| AssayKitCatalogueNumber |
| Description |
The catalogue/product number of the assay kit |
| Field Type |
Recommended |
| Data Type |
Text |
| Examples |
MET-5163, 23227 |
| AssayKitLotNumber |
| Description |
The lot/control number of the assay kit |
| Field Type |
Recommended |
| Data Type |
Text |
| AssayReagent1 |
| Description |
If not using an assay kit, indicate the common name of the reagent used for the assay.
Reagent is a substance or mixture for use in cell culture media, chemical analysis or other reactions. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| AssayReagent1Concentration |
| Description |
Concentration of the working reagent.
Definition: a solution that is prepared for immediate use in the analytical procedure, often by combining a stock solution (or multiple reagents) with a diluent. |
| Field Type |
Conditionally required |
| Data Type |
Numeric |
| AssayReagent1Unit |
| Description |
Unit of the concentration of the working assay reagent, preferably g/mL |
| Field Type |
Conditionally required |
| Data Type |
Text |
| BufferName |
| Description |
Common name of the buffer used in the experiment. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| BufferConcentration |
| Description |
Concentration of the working solution of the buffer. |
| Field Type |
Conditionally required |
| Data Type |
Numeric |
| BufferConcentrationUnit |
| Description |
Unit of the concentration of the working buffer solution, preferably Molar concentrations. |
| Field Type |
Conditionally required |
| Data Type |
text |
| OECDTGD |
| Description |
Name and number of OECD Technical Guidance Document if followed for conducting the experiment. |
| Field Type |
Conditionally required |
| Data Type |
text |
| Examples |
OECD Test Guideline number 471: Bacterial Reverse Mutation Test |
| OtherGD |
| Description |
Details of any other guidance document (i.e., non-OECD) being followed. Provide as much detail as possible. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
ASTM E1439-12: Standard Guide for Conducting the Frog Embryo Teratogenesis Assay-Xenopus (FETAX) |
| Protocol |
| Description |
Title of the experimental protocol.If multiple protocols exist, please list them e.g. sample preparation protocol, sequencing protocol |
| Field Type |
Recommended |
| Data Type |
Text |
| ProtocolURL |
| Description |
The weblink of the Standard Operating Procedure. Initially MS-Teams link can be shared, once finalised can be uploaded to protocols.io (https://www.protocols.io/welcome-springernature) |
| Field Type |
Conditionally required |
| Data Type |
Text |
| ManufacturerInstrumentName |
| Description |
Name of the manufacturer followed by instrument name |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
ThermoFisher Scientific - Varioskan ALF |
| TypeOfMeasurement |
| Description |
Detail the type of analytical measurement the instrument does |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
spectrometry (nm), fluorometry (nm) |
| PositiveControl |
| Description |
Common name of the compound that is used as a positive control in the assay. If positive control is not used, mention ‘not used’.
Definition: Separate part of the test system treated with an item for which it is known that the test system should respond. The positive control provides evidence that the test system is responsive under the actual conditions of the assay. The positive control is endpoint specific to the test system (ref: GIVIMP, Second Edition) |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Sodium Dodecyl Sulphate, Lipopolysaccharide |
| PositiveControlConcentration |
| Description |
The concentration of the compound used as a positive control |
| Field Type |
Conditionally required |
| Data Type |
Numeric |
| PositiveControlUnit |
| Description |
The measurement unit of concentration of the compound used as a positive control, in mM or mg/L |
| Field Type |
Conditionally required |
| Data Type |
Text |
| NegativeControlType |
| Description |
Definition: Separate part of a test system treated with an item for which it is known that the test system should not respond; the negative control provides evidence that the test system is not responsive under the actual conditions of the assay (ref: GIVIMP, Second Edition)
Solvent/vehicle control (consist of solvent or vehicle alone, without test item (test material), and otherwise treated in the same way as the treatment groups), untreated control (consist of culture medium without solvent / vehicle or test item and otherwise treated in the same way as the treatment groups), true negative control (items (e.g. chemicals) with known lack of activity). Ref: OECD 201
If any type of negative control is not used, mention ‘not used’ |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Select from dropdown |
| NegativeControl |
| Description |
Describe the negative control.
. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| NegativeControlConcentration |
| Description |
The concentration of the compound used as a negative control |
| Field Type |
Conditionally required |
| Data Type |
Numeric |
| NegativeControlUnit |
| Description |
The measurement unit of concentration of the compound used as a negative control, in mM or mg/L |
| Field Type |
Conditionally required |
| Data Type |
Text |
| NumberOfBiologicalReplicates |
| Description |
The number of biological replicates.
Biological replicates are parallel measurements of biologically distinct samples that capture random biological variation, which may itself be a subject of study or a noise source.
Ref: Blainey, P., Krzywinski, M., Altman, N., 2014. Replication. Nature Methods 11, 879–880. https://doi.org/10.1038/nmeth.3091 |
| Field Type |
Recommended |
| Data Type |
Integer |
| NumberOfTechnicalReplicatesPerCondition |
| Description |
The number of technical replicates per concentration (single, duplicate, triplicate)
Technical replicates are repeated measurements of the same sample that represent independent measures of the random noise associated with protocols or equipment.
Ref: Blainey, P., Krzywinski, M., Altman, N., 2014. Replication. Nature Methods 11, 879–880. https://doi.org/10.1038/nmeth.3091 |
| Field Type |
Recommended |
| Data Type |
Integer |
| DataAcquisition |
| Description |
The software/platform, if any, used for data acquisition |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
MassHunter Workstation (version B.07.00, Agilent);BD FACSDiva™ Software |
| RawDataProcessingSteps |
| Description |
The steps for processing raw data.
Data processing workflow can be created and submitted in https://workflowhub.eu |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Normalisation to negative/positive/vehicle control/ blank corrections, use of calibration curve, subtraction of control values, calculation of averages, standard deviations. Mention the statistical methods used to derive the parameters to be reported. |
| SoftwareForDataProcessing |
| Description |
The name of the software (and version) for processing raw data. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
If a software is not used, mention “not used”
Microsoft® Excel® for Microsoft 365 MSO (Version 2512 Build 16.0.19530.20038) 64-bit, GraphPadPrism (GraphPad Software, La Jolla, CA, USA) |
| SoftwareSettingsForDataProcessing |
| Description |
Configuration and parameters defined within a software tool (other than Microsoft Excel) to analyse raw experimental data. |
| Field Type |
Conditionally required |
| Data Type |
Text |
| Examples |
Format for raw data (fluorescence intensity, absorbance values, etc.) à thresholds/cutoff values for detecting significant changes à statistical tests à etc. |
| OutlierHandling |
| Description |
Explanation of how outliers are handled, i.e., what (statistical) method has been used to exclude values. |
| Field Type |
Recommended |
| Data Type |
Text |
| SoftwareToDetermineEC50 |
| Description |
Name of the software used to determine half maximal inhibition or effect concentration (if used) |
| Field Type |
Conditionally required |
| Data Type |
text |
| Examples |
GraphPad Prism, ProTox-3.0 |
| Researcher |
| Description |
The role a person has of being a researcher involved in a research investigation, for example by conducting experiments, collecting and analysing data, or undertaking formal study (ref: http://purl.org/spar/scoro/researcher)
In addition to credit and provenance, it helps in quality control and data tracking. Provide full name of the person, preferably as registered in ORCID registry, or name is scientific publications if any, or professional name most widely used. |
| Field Type |
Mandatory |
| Data Type |
text |
| Examples |
Jane Doe |
| ResearcherORCID |
| Description |
ORCID ID of the person responsible for conducting the research and investigation process, specifically performing the experiments, or data/evidence collection, or the person who created the resource. In addition to credit and provenance, it helps in quality control and data tracking. |
| Field Type |
Mandatory |
| Data Type |
String/integer |
| Examples |
0000-0002-6125-0555 |
| DescriptionOfDataGeneration |
| Description |
Explain the datasets that would be generated from the studies
Dataset: Organised collection of data or objects in a computational format, that are generated or collected by researchers in the course of their investigations, regardless of their form or method, that form the object on which researchers test a hypothesis. This includes the full range of data: raw, unprocessed datasets, proprietary generated and processed data and secondary data obtained from third parties. Ref: https://terms.codata.org/rdmt/dataset
This question is also in PARC DSW Data Management Plan tool. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Data related to relative (fold change) in hormone levels at each concentration, normalised metabolite ratio peak data per sample |
| SummaryDatafileReference |
| Description |
Name of the file which contains measured biological response results. |
| Field Type |
Recommended |
| Data Type |
String |
| Examples |
Stringsummary.csv; stringsummary.xlsx |
| Filestoragelocation |
| Description |
The location where the file is stored |
| Field Type |
Mandatory |
| Data Type |
String |
| Examples |
University repository, Research Data Storage options provided by institutions, SharePoint, OneDrive. |
Assay Summary Result sheet
| ExperimentID |
| Description |
The experiment ID – the sytax to be decided by WP5. Can be extrapolated from the the MethodNameID.Used for internal reference. |
| Field Type |
Recommended |
| Data Type |
String |
| TestSubstance |
| Description |
Common/widely used name of the chemical/substance being tested |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Bisphenol Z |
| SampleID |
| Description |
The sample ID of the experiment; syntax to be finalised by WP5. A possible syntax can be MethodNameID- experiment ID- sample number |
| Field Type |
Mandatory |
| Data Type |
String |
| TestSubst anceID |
| Description |
An identifier given in the project. |
| Field Type |
Recommended |
| Data Type |
String |
| CellLine |
| Description |
A cell line is defined as a genetically stable and homogenous population of cultured cells that shares a common propagation history (i.e. has been successively passaged together in culture). Ref: Sarntivijai, Sirarat, et al. 2014. “CLO: The Cell Line Ontology.” Journal of Biomedical Semantics 5(1):37. doi: 10.1186/2041-1480-5-37. |
| Field Type |
Mandatory |
| Data Type |
Text, URL |
| Examples |
HepG2, A549, hiPS - Human induced-pluripotent stem cells, 293FT,Human embryonic stem cell (hESC) |
| CultureMediumComposition |
| Description |
Describe the cell culture medium composition briefly with key components, such as serum type and percentage, antibiotics. |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Roswell Park Memorial Institute Medium (RPMI 1640) supplemented with 10 % (v/v) Fetal Bovine Serum (FBS) heat-inactivated, 100 U/mL penicillin, 100 μg/mL streptomycin, and 4 mM/L glutamine |
| SerumPercentage |
| Description |
Percentage of serum used in the cell culture media. Avoid adding percentage symbol after reporting the data. |
| Field Type |
Recommended |
| Data Type |
Numeric |
| HeatInactivation |
| Description |
Whether the serum was heat activated or not as “yes” or “no” response |
| Field Type |
Mandatory |
| Data Type |
Text |
| Examples |
Yes, No |
| NumberOfBiologicalReplicates |
| Description |
The number of biological replicates.
Biological replicates are parallel measurements of biologically distinct samples that capture random biological variation, which may itself be a subject of study or a noise source.
Ref: Blainey, P., Krzywinski, M., Altman, N., 2014. Replication. Nature Methods 11, 879–880. https://doi.org/10.1038/nmeth.3091 |
| Field Type |
Recommended |
| Data Type |
Integer |
| NumberOfTechnicalReplicatesPerCondition |
| Description |
The number of technical replicates per concentration (single, duplicate, triplicate)
Technical replicates are repeated measurements of the same sample that represent independent measures of the random noise associated with protocols or equipment.
Ref: Blainey, P., Krzywinski, M., Altman, N., 2014. Replication. Nature Methods 11, 879–880. https://doi.org/10.1038/nmeth.3091 |
| Field Type |
Recommended |
| Data Type |
Integer |
| ExposureConcentrations |
| Description |
The measured (if not measured, then nominal) amount(s)of the test substance used in the exposure scheme. |
| Field Type |
Mandatory |
| Data Type |
Numeric |
| ExposureConcentrationUnit |
| Description |
Unit of the test substance’s measured/nominal concentration (preferably Molar concentrations). Please do not use special characters like µ to avoid mojibake. |
| Field Type |
Mandatory |
| Data Type |
Text |
| ExposureDuration |
| Description |
The total time of exposure to the test substance |
| Field Type |
Mandatory |
| Data Type |
Integer |
| Examples |
24, 48, 7 |
| UnitofExposureDuration |
| Description |
Unit of exposure time/duration |
| Field Type |
Mandatory |
| Data Type |
text |
| Examples |
hrs, d |
| RecoveryPeriod |
| Description |
Describe the treatment free period |
| Field Type |
Recommended |
| Data Type |
Text |
| PositiveControlEffect |
| Description |
Response of test system to positive control |
| Field Type |
Mandatory |
| Data Type |
Text |
| ResponseOfTestSystemNormalised |
| Description |
Response of the test system/cell line |
| Field Type |
Mandatory |
| Data Type |
Numeric |
| Examples |
IC50, EC50, EC20, BMC50, log2_fold_induction, log10_fold_induction, Percent activity |
| KeyEventID |
| Description |
If available, add the Key Event ID from AOPWiki. |
| Field Type |
Optional |
| Data Type |
Text |
| Examples |
KE:2067 |
| Comments |
| Description |
Add comments regarding worth mentioning points not captured earlier.e.g., reasons for data exclusion, consideration of cytotoxicity or other type of interference that can impact the results |
| Field Type |
Optional |
| Data Type |
Text |